Real time case studies
At Accumold, we use micro AM as a tool to reduce the time from initial design to market. When looking at a conventional prototyping scenario, prototype tooling needs to be fabricated at often considerable cost, and also taking upwards of 6 weeks, meaning that initial testing cannot take place until at least week 7 of a product development process. The time involved when using conventional tooling is only exacerbated if (as is often the case) design changes are required, and tooling needs to be modified.
When using micro AM, by week 7 a design can have been refined numerous times as the process requires no traditional tooling, just DRST. This means that time and focus in this early stage of product development can be spent on design optimisation, and so micro AM not only saves time and cost in product development, but it also stimulates innovation.
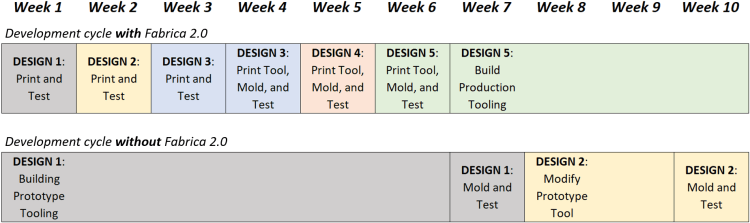
Table 1. A representation of how the use of micro AM in a product development cycle can significantly reduce time to market which in itself reduces product development costs and stimulates design optimization.
The theoretical case for the use of micro AM in precision plastics part and component development is seemingly therefore extremely compelling. However, Accumold now has significant experience in using the technology for customers, and so we are able to illustrate the way in which micro AM has benefitted OEMS in real time.
Due to confidentiality issues related to the case studies discussed below, we are unable to include pictures of the products discussed. Pictures used in this article are non-related part shots that show the precision and resolution that can be achieved using the Fabrica 2.0 technology.
Case Study 1 - Intraocular Implant
Our first example is a medical OEM customer making an intraocular implant that measured 1.6 x 2.9 x 0.7 mm. The customer was up against a looming FDA submission deadline. The part design included a small gap on one side of the part. While the rest of the part design was pretty much frozen, the customer needed to determine what gap would be optimal for the part considering that in its final assembly it would need to be accurately handled and safely implanted inside the eye.
Due to the micro of this part, using the Fabrica 2.0 we could print a lot of samples in the 50mm x 50mm x 100mm print envelope of the machine. We successfully printed multiple samples of six different designs for the customer to test. This took less than 24 hours to design, print, post process, package, and ship out to the customer, which was able to complete their testing in about two weeks. Their tests proved one of the designs superior to all others, and this was the design the company froze and submitted for FDA approval. Had we not been able to provide multiple part iterations for testing at one time the customer would have had to delay their FDA submission or choose a design without adequate test data. Once the customer is satisfied with their test performance Accumold can start to design and build the production tool so that we can qualify the tool and scale up to meet production volumes.

Micro gears made using the Nano-Dimension Fabrica 2.0 micro AM machine. The smallest gears shown have 20 teeth contained on a wheel diameter of 1.25 mm, each tooth measuring 100 microns.


